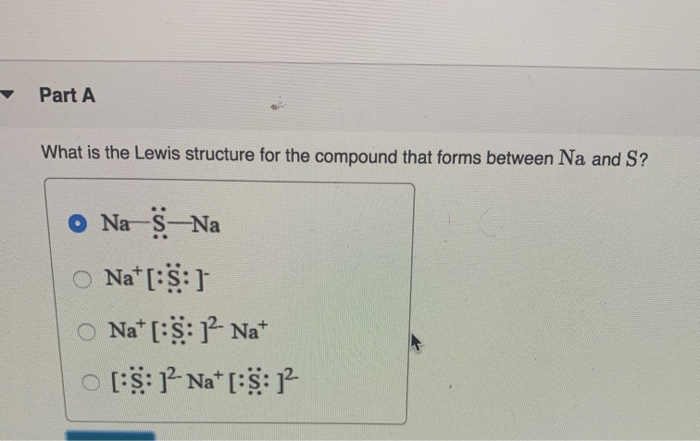
Sodium Sulfide (Na2S)- Structure, Molecular mass, Chemical properties, Physical Properties with Uses and FAQs of Sodium Sulfide.

ENa^ + /Na(s)^0 = - 2.71V , EMg^2 + /Mg(s)^0 = - 2.37V , EFe^2 + /Fe(s)^0 = - 0.44V and ECr^3 + /Cr(s)^0 = - 0.41V Based on this data, state which of the following is the weakest reducing agent.